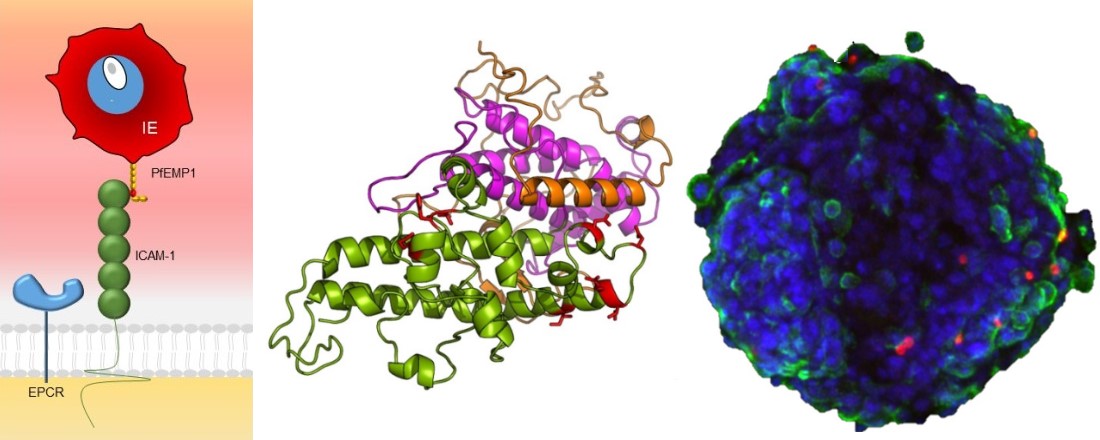
Malaria immunity targets team (TARGETS)
Research in the TARGETS team is focused on a family of "sticky" proteins known as the Plasmodium falciparum erythrocyte membrane protein 1 (PfEMP1). P. falciparum is the most pathogenic malaria parasite and a major cause of morbidity and mortality among children in sub-Saharan Africa.
Our current research projects include in vitro molecular and immunological characterization of PfEMP1 proteins associated with cerebral malaria in young children.